TAP Psychopharmacology Bulletin - New Horizons / 2025 July
Prepared for clinicians and researchers working in the field of psychiatry.
Bulletin Subscription
Be the first to know when new issues are published! Apply now for free subscription.
Free • Cancel anytime you want
News
Semaglutide-Induced Lithium Toxicity
One of the most common challenges encountered in clinical psychiatric practice is weight gain associated with psychotropic medications and the subsequent complications arising as a result of it. These complications are not only limited to changes in physical appearance and also encompass significant health risks to the patient.
Although various pharmacological and non-pharmacological strategies have been employed to manage weight gain, this issue remains prevalent and cannot be overlooked by psychiatric services. Novel treatments for obesity offer promising alternatives, particularly for overweight individuals and patients experiencing drug-induced weight gain.
As with any pharmacological intervention, the benefit-risk profile of these treatments must be carefully evaluated. Semaglutide, a glucagon-like peptide-1 (GLP-1) receptor agonist, recently emerged as a treatment option for obesity. It exerts its effects primarily by delaying gastric emptying. Nevertheless, potential drug interactions and clinical outcomes associated with semaglutide are not fully elucidated yet. Consequently, sharing clinical experiences from real-world settings is critical in guiding other clinicians.
- Aripiprazole, Brexpiprazole, and Cariprazine
- First-Episode Schizophrenia and Prolonged D2 Blockade
- Neurosteroid-based Treatments in Postpartum Depression
- A New Generation Approach in the Treatment of Schizophrenia: NBI-1117568
- A New Molecule: Lurasidone in Children and Adolescents
Development of lithium toxicity in a patient who had been using oral lithium at a stable dose of 1200 mg/day for 14 months with therapeutic serum levels and no signs of toxicity. Following the initiation of semaglutide treatment, the patient's serum lithium level increased from 0.9 mEq/L to 2.4 mEq/L, despite no change in the lithium dosage. The lithium level returned to normal with intravenous hydration, and the therapeutic serum level was subsequently maintained with a reduced lithium dose of 600 mg/day.
GLP-1 receptor agonists enhance insulin secretion while suppressing glucagon and delaying gastric emptying in a dose-dependent manner. This delayed gastric transit may interfere with the absorption and distribution of concurrently administered oral medications. In conclusion, while new pharmacological agents such as semaglutide hold promise in the treatment of conditions like obesity, clinicians must remain vigilant regarding possible interactions with ongoing psychotropic regimens. Careful monitoring and individualized assessment are essential to ensure safe and effective treatment outcomes.
M.D. Furkan Yazıcı
DOI: 10.1097/JCP.0000000000002017
DOI: 10.1007/5584_2020_496
Does AI Outperform Psychiatrists in Tardive Dyskinesia Screening?
One of the major complications associated with the long-term use of antipsychotic medications in clinical practice is tardive dyskinesia (TD). However, TD is often overlooked or diagnosed late. In the United States, this motor disorder affects approximately 2.6 million individuals, yet only about 40,000 patients receive treatment. Early diagnosis plays a crucial role in preventing the progression of this condition. Although there are approved treatments for TD such as valbenazine and tetrabenazine,
delayed diagnosis often limits access to and effectiveness of these therapies. With the integration of artificial intelligence (AI)-based tools into clinical practice, it is anticipated that the quality of life for patients may improve and the burden on the healthcare system may also be reduced. A recently published studydemonstrated that video-based AI technologies hold promising potential in this area. In a three-phase study design, short video recordings of patients using antipsychotic medications were analyzed andTD symptoms were assessed using an algorithm that evaluated facial, shoulder, and hand movements. The study reported noteworthy findings.
Compared to human raters, the AI- supported model demonstrated higher sensitivity and specificity, with a mean AUC (area under the curve) of 0.89, indicating a high capacity in detecting TD.
Although the model is not designed to replace clinical diagnosis, researchers suggest it may be used as a screening tool, particularly in telepsychiatry, by identifying high-risk patients early and saving clinicians time.
M.D. Gözde Çolak
DOI: 10.4088/JCP.25m15792
From the Editor...

Dear Colleagues, As we celebrate the 20th anniversary of the Psychopharmacology Association, it is my pleasure to introduce the Current Perspectives in Psychopharmacology bulletin, which we have launched with the aim of sharing the latest developments in our field.
The journey of psychiatric pharmacology began in the 1950s and has gained significant momentum in recent years, particularly with the contribution of artificial intelligence technologies. Today, molecules can be designed virtually and data from previous patients allow us to predict the efficacy and side effects of new medications. Many developments that were considered science fiction only a few years ago have now become a part of our daily clinical practice. This rapid transformation brings with it a responsibility to stay informed about innovations in our field. To address this need, we have initiated this quarterly bulletin—entirely independent of the pharmaceutical industry—under the auspices of the Psychopharmacology Association. Our goal is to provide clear, concise, and up-to-date content that you DOI: 10.1176/appi.ajp.20240321 can easily browse during short breaks in your busy clinical schedules. We believe the visual materials included will enhance memory retention and improve the accessibility of the content.
Turkey was once a significant stakeholder in the international pharmaceutical industry during the 1990s and early 2000s. However, this has changed in recent years. Obtaining marketing authorization for new medications now takes considerable time, and even access to generic drugs can occasionally pose challenges. Several factors and policy decisions underlie this shift.
Understandably, these developments directly affect many of our members, the majority of whom are actively engaged in patient care. The most effective way to navigate these challenges is to adopt a proactive approach.
In this context, we also plan to include brief news and commentaries addressing current treatment challenges and potential solutions. Your clinical insights and contributions will greatly enhance the relevance and impact of this bulletin. We warmly welcome contributions from all colleagues who wish to support this initiative. Your input will help make this publication more comprehensive, inclusive, and useful for us all.
Best regards,

Is Selenium Neuroprotective?
In recent years, the search for effective treatments in neurodegenerative diseases has accelerated, with numerous agents being tested, particularly in rodent models. In a study aiming to enhance neuronal recovery following a stroke, selenium treatment was shown to significantly reduce cerebral infarct volume and oxidative stress in rats subjected to transient cerebral artery occlusion. It also demonstrated improved motor performance in rats during the acute phase post-stroke.
Selenium is recognized as a trace element and a critical antioxidant. A growing body of evidence has confirmed its neuroprotective properties against various neurotoxic agents. It has been well established that long-term lithium treatment, commonly used in psychiatric disorders, may lead to multi-organ dysfunction, particularly affecting the nervous system. Cerebellar toxicity is a rare but serious example of the adverse effects of lithium treatment. In a rat model of lithium toxicity, selenium administration was found to reduce oxidative stress markers, attenuate lithium-induced cerebellar alterations, increase Klotho expression, and decrease the expression of caspase-3 and GFAP (glial fibrillary acidic protein).
Furthermore, a meta-analysis evaluating clinical studies on Alzheimer's disease revealed that selenium supplementation improved glutathione peroxidase (GPX) activity and performance in certain cognitive tests in patients with mild cognitive impairment. Mini-Mental State Examination (MMSE) scores in patients with Alzheimer's disease also improved. In conclusion, selenium supplementation may be considered a potential adjunctive therapy in psychiatric and neurological disorders with a neurodegenerative component.
DOI: 10.1177/0271678X231156981
DOI: 10.1016/j.tice.2025.102779
DOI: 10.3390/nu14153205
- Prof. Dr. M. Kemal SAYAR
- Prof. Dr. Mesut ÇETİN
- Prof. Dr. Feyza ARICIOĞLU
- Prof. Dr. Ali Saffet Gönül
- M.D. Gözde Çolak
- Yiğit Erdoğan
Erenkoy District Kadikoy, Istanbul 34738
psychopharmaupdate@gmail.com
Its content is independent of the pharmaceutical industry.
ARIPIPRAZOLE, BREXPIPRAZOLE, AND CARIPRAZINE
THREE DIFFERENT SISTERS
With the recent introduction of cariprazine to the antipsychotic (AP) market in Turkey, the number of APs exhibiting partial agonist properties has increased to three. Although aripiprazole (ARI), brexpiprazole (BRX), and cariprazine (CAR) share a common mechanism of action, they differ significantly in terms of pharmacokinetics, pharmacodynamics, administration patterns, side effect profiles, and patient tolerability. Understanding these differences allows for a more rational and individualized antipsychotic selection in clinical practice.
Dopamine Receptor Blockade and Clinical Implications
A series of PET studies led by Kapur and Seeman demonstrated that approximately 65–75% D2 receptor occupancy in the basal ganglia is sufficient to elicit an antipsychotic response. Beyond this threshold, there is a significantly increased risk of motor side effects and elevated prolactin levels. Long before these findings, some astute clinicians had proposed that antipsychotic efficacy could be achieved without the emergence of motor side effects, and that such side effects may indicate overdosing; however, this hypothesis lacked robust empirical support and validation. In everyday clinical practice, prescribing antipsychotics without inducing adverse side effects poses a major challenge, particularly due to concerns about subtherapeutic dosing. In this context, newer antipsychotics with partial agonist activity—often referred to as “smart drugs” —have gained prominence. ARI, BRX, and CAR are three key representatives of this class.
Aripiprazole (ARI)
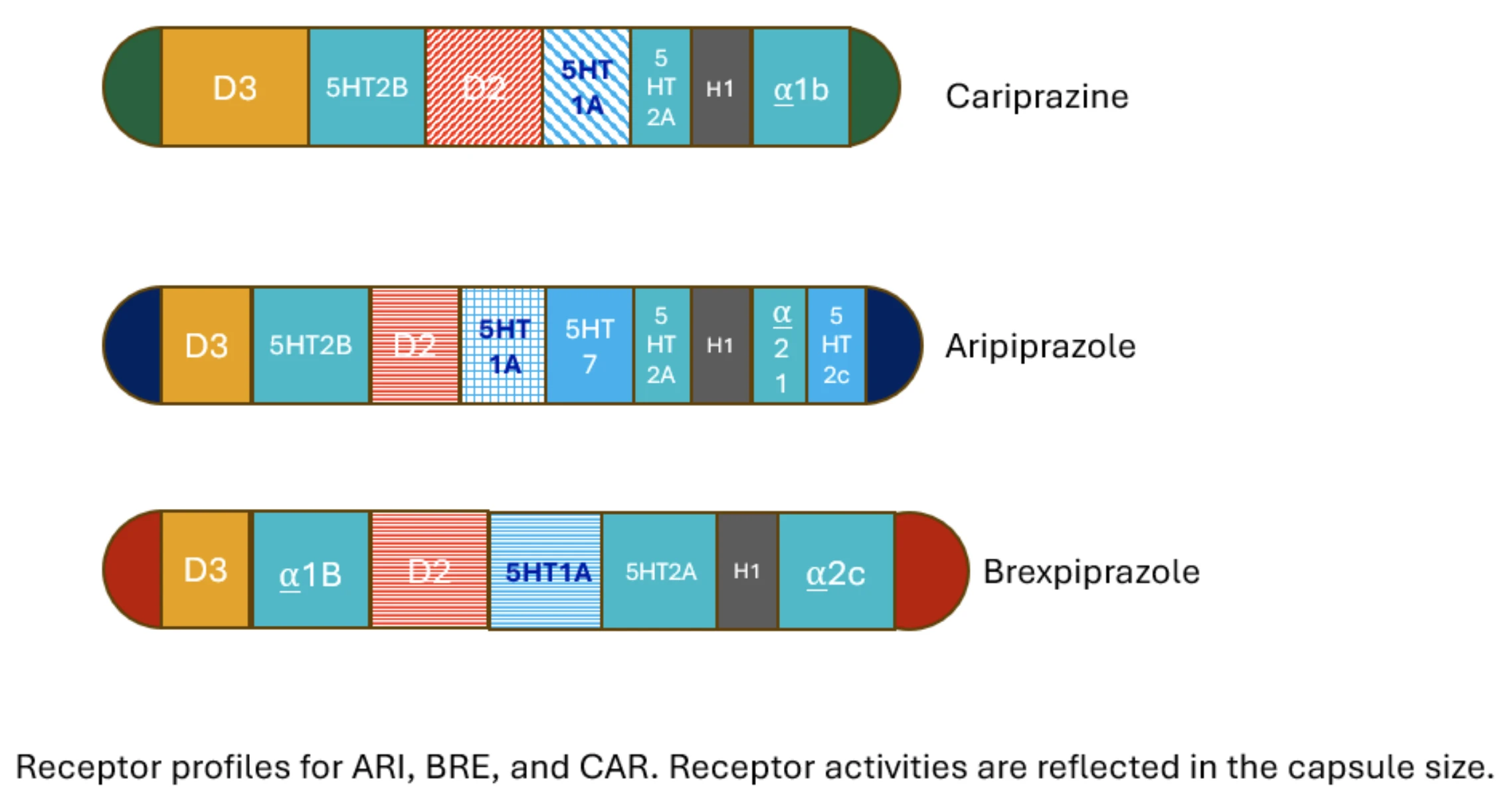
Unlike conventional antipsychotics, aripiprazole not only blocks D2 receptors, but also acts as a partial agonist, maintaining a degree of dopaminergic activity (Eₘₐₓ vs. dopamine ≈ 50–60%). This allows for the preservation of approximately 25–30% of physiological dopamine function, which is thought to prevent side effects such as hyperprolactinemia and parkinsonism. Of note, 1mg of ARI occupies approximately 50% of postsynaptic D2Rs, while 10 mg can occupy up to 95%.
Concerns that antipsychotics may not exert sufficient clinical effects at subtherapeutic doses often lead to the use of unnecessarily high doses in treatment, thereby increasing the risk of undesirable side effects.
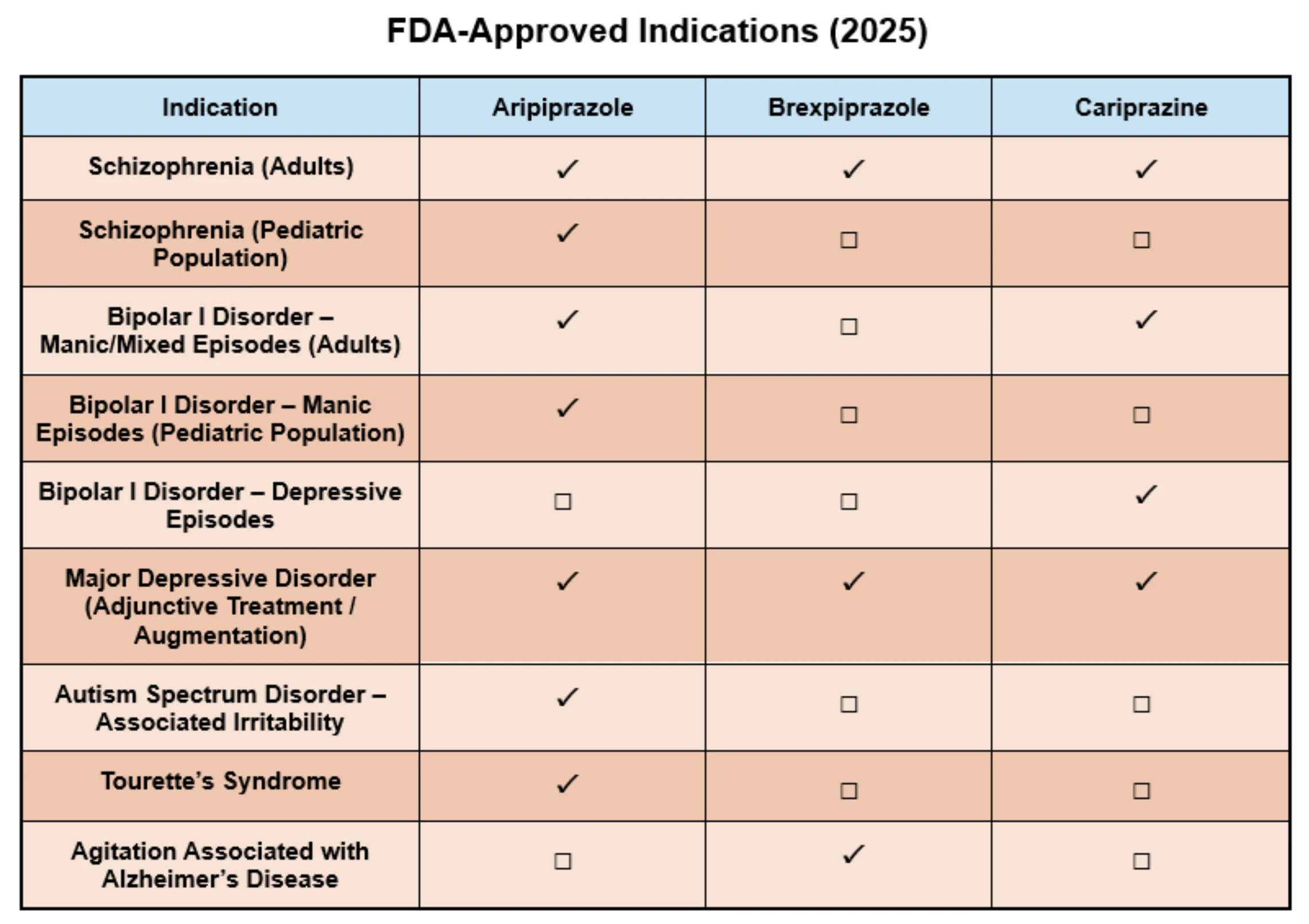
Clinical observations have largely confirmed this theoretical benefit: increases in prolactin levels and motor side effects are rare, whereas akathisia and restlessness are prominent side effects. Akathisia is believed to result from dopaminergic stimulation due to partial agonism and is reported in approximately 8% of schizophrenia patients as well as up to 25% of patients with depression. Since 2002, ARI has received FDA approval for multiple psychiatric disorders in both oral and long-acting injectable (LAI) forms (see Table 1).
Brexpiprazole (BRX)
Brexpiprazole exhibits lower intrinsic dopaminergic activity compared to ARI (Eₘₐₓ ≈ 43%), mimicking approximately 20% of physiological dopamine signaling. This reduced activity is associated with a lower risk of akathisia. Brexpiprazole exhibits lower intrinsic dopaminergic activity compared to ARI (Eₘₐₓ ≈ 43%), mimicking approximately 20% of physiological dopamine signaling. This reduced activity is associated with a lower risk of akathisia. In addition, BRX has more prominent 5-HT1A partial agonist, 5-HT2A antagonist, and alpha1B antagonist properties than ARI, potentially reducing side effects such as anxiety and agitation. A 4 mg dose of BRX blocks around 80% of postsynaptic receptors, accounting for approximately 20% residual activity due to partial agonism.
Cariprazine (CAR)
Cariprazine stands apart pharmacodynamically due to its strong affinity not only for D2 receptors—similar to BRX—but also for D3 receptors. It exhibits approximately 40% intrinsic activity at D2Rs and up to 60% at D3Rs. At a daily dose of 3 mg, CAR binds ~92% of D2Rs and ~79% of D3Rs. Notably, CAR may offer benefits for primary negative symptoms in schizophrenia, likely due to its high D3 affinity. D3 autoreceptor blockade in the ventral tegmental area may enhance dopamine release in the prefrontal cortex, which could improve negative symptoms and contribute to its antidepressant effects. Consequently, CAR is the only dopamine partial agonist approved for bipolar depression (NNT = 4–10).
Comparative Clinical Efficacy
All three agents share partial 5- HT1A agonist, 5-HT2A antagonist, and partial D2 agonist properties, which are believed to augment antidepressant effects in major depressive disorder (MDD).
Estimated NNT values are as follows: ARI = 9, BRX = 12–16, CAR = 9–16. The antidepressant effect of CAR is thought to benefit from its D3R partial agonism and full 5-HT1A agonist activity.
All three drugs are effective in the treatment of acute schizophrenia. Comparative efficacy data suggest similar outcomes with NNTs as follows: ARI = 8, BRX = 7, CAR = 10. For relapse prevention during maintenance therapy, NNT values are reported to be between 4 and 5 for all three agents.
Pharmacokinetics, Drug Interactions, and Metabolism
Pharmacokinetic profiles differ considerably among the three agents. ARI has a half-life of approximately 75 hours, and with its active metabolite, dehydro-aripiprazole, total elimination may extend to 86 hours. BRX has a half-life of approximately 94 hours and lacks a major active metabolite. CAR, on the other hand, has a variable half-life of 2–4 days, with active metabolites that may take 1–3 weeks for full elimination. This extended profile may delay steady-state levels, leading some clinicians to prematurely increase doses due to a perceived lack of early efficacy. Nevertheless, the long half-life and active metabolites provide a buffer against missed doses —an advantage in clinical practice.
All three drugs are primarily metabolized via CYP2D6 and CYP1A2.
Agents that inhibit (e.g., fluoxetine, duloxetine, risperidone, paroxetine, propranolol) or induce (e.g., carbamazepine) these enzymes can significantly alter both the parent drug and metabolite exposure durations.
Conclusion and Clinical Implications
In summary, while aripiprazole, brexpiprazole, and cariprazine share a common pharmacological mechanism as dopamine partial agonists, they differ substantially in pharmacodynamic and pharmacokinetic profiles, approved indications, and side effect tolerability.
These "three sisters" offer effective treatment options across the schizophrenia and mood disorder spectrum. A thorough understanding of their distinct biological and clinical features is crucial for personalized treatment selection and optimized side effect management.
FIRST-EPISODE SCHIZOPHRENIA AND PROLONGED D2 BLOCKADE:
A NECESSİTY OR AN OVERTREATMENT?

Introduction
The long-term use of antipsychotics in the treatment of schizophrenia has been a subject of ongoing debate for years. One key point of discussion is whether sustained dopamine D2 receptor blockade remains effective over time. A study published in 2025 by Tiihonen et al. in the American Journal of Psychiatry, focused specifically on first-episode schizophrenia patients who were maintained under continuous D2 blockade and followed for up to 10 years to assess relapse rates.
Study Design and Methodology
Using Finland's national health registry, individuals under the age of 45 who were hospitalized with a firsttime diagnosis of schizophrenia between 1996 and 2014 were screened. A total of 4,788 patients with no prior antipsychotic exposure were included in the study. Of these, 305 patients received long-acting injectable (LAI) antipsychotics within 30 days of diagnosis, forming the “continuous treatment” group. The remaining patients, who were either untreated, intermittently treated, or received oral antipsychotics, constituted the “other” group. The primary outcome was defined as hospitalization due to psychotic relapse, with a follow-up duration of 10 years.
This study demonstrates that sustained D2 blockade is effective and safe in the treatment of schizophrenia. It shows a reduction in relapse rates among patients using LAIs.
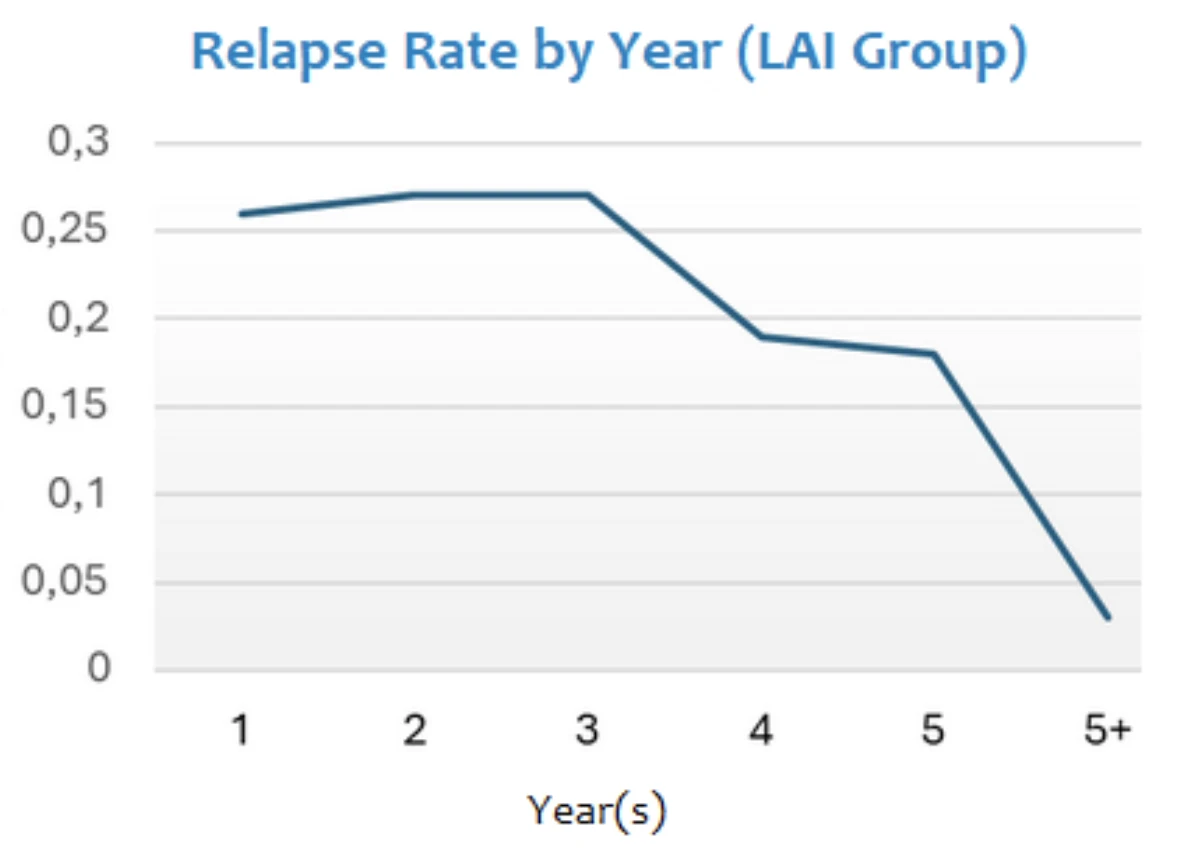
Results – Relapse Rates and Trends
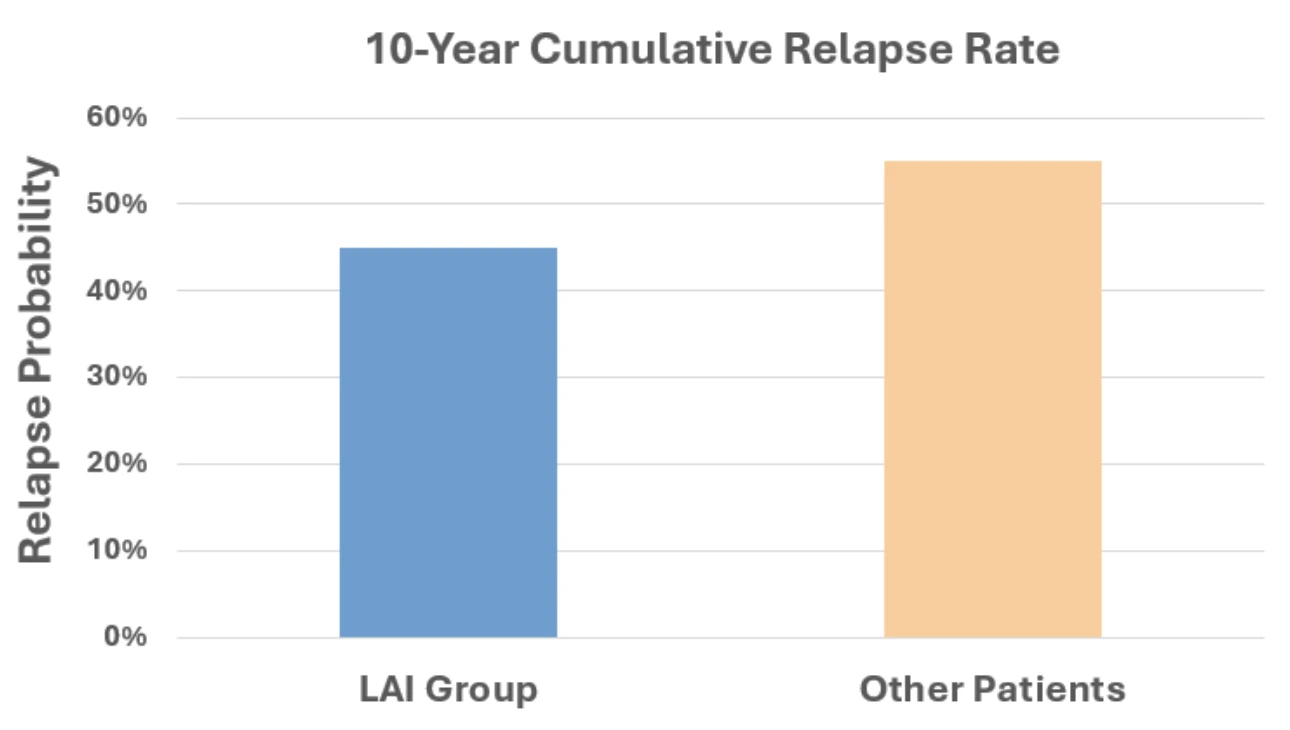
In the LAI group (n = 305), a 45% relapse rate was observed over 10 years, corresponding to a 55% relapse-free rate. The annual relapse rate per person-year was 26% in the first year, which declined to 5% by year five. From the sixth year onward, only four relapses occurred, reflecting a 3% annual relapse rate. Relapse rates decreased significantly with continued treatment. In contrast, the “other” treatment group had a 55% relapse rate over 10 years.
Response to the Dopamine Supersensitivity Hypothesis
The dopamine supersensitivity hypothesis posits that prolonged D2 blockade may sensitize dopamine receptors, potentially leading to treatment-resistant psychotic episodes over time.
However, this study found the opposite trend: as treatment duration increased, relapse rates decreased, no dose escalation was needed, and in fact, median DDD (defined daily dose) declined from 1.20 to 1.04 over the 10-year period.
Clinical Implications
1. Early initiation of LAIs after the first episode may reduce relapse rates.
2. The dopamine supersensitivity theory is not supported by long-term data.
3. Improved treatment adherence through LAIs may contribute to long-term clinical stability.
4. Preventing relapse is critical, as each episode may lead to a decline in treatment responsiveness.
5. Preventing relapse is critical, as each episode may lead to a decline in treatment responsiveness.
Conclusion and Commentary
This study demonstrates that continuous D2 receptor blockade is both effective and safe in the longterm treatment of schizophrenia. In patients receiving LAIs, relapse rates decreased over time, and treatment efficacy was maintained. These findings challenge the dopamine supersensitivity hypothesis and support sustained dopamine blockade as a central strategy for relapse prevention and long-term functional recovery in schizophrenia.
NEUROSTEROID-BASED TREATMENTS IN POSTPARTUM DEPRESSION:
BREXANOLONE AND ORAL ALTERNATIVES

Postpartum depression (PPD) affects approximately 10–15% of women and is characterized by significant mood symptoms and functional impairment. The therapeutic effects of traditional antidepressants typically emerge within 2–4 weeks. However, in the context of PPD— particularly regarding early intervention and mother-infant bonding—faster-acting agents are needed. Neurosteroid-based treatments have garnered increasing attention in this regard. Brexanolone, a GABA-A receptor modulator, along with its oral alternatives, have begun to reshape the treatment paradigm in recent years.
IV Brexanolone: The First and Only Approved Intravenous Treatment
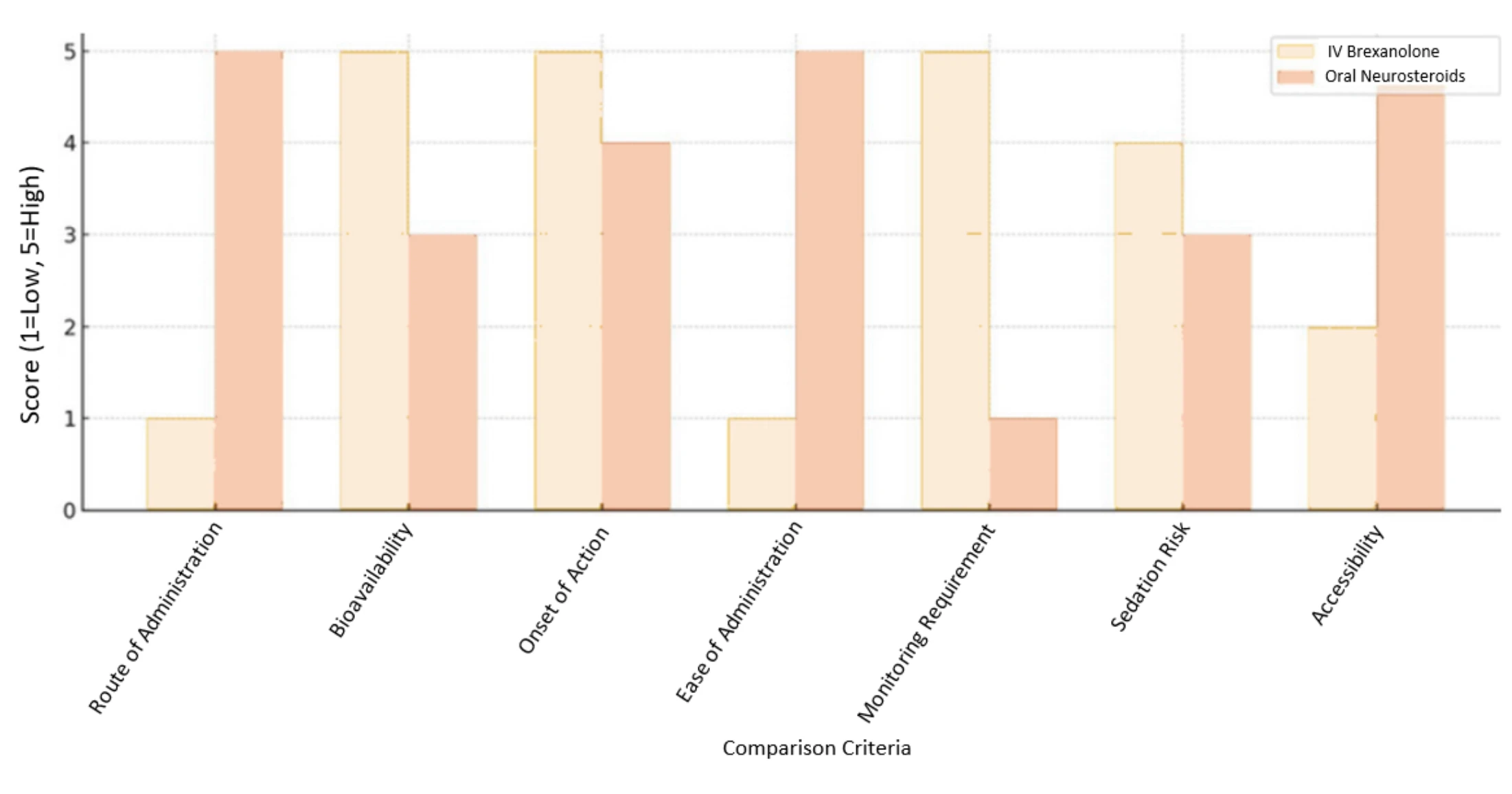
Brexanolone (Zulresso®) is the intravenous formulation of allopregnanolone. In 2019, it became the first FDA-approved medication specifically indicated for the treatment of PPD. Its mechanism of action involves positive allosteric modulation of GABA-A receptors, resulting in a rapid and robust therapeutic effect.
Phase III clinical trials—namely the ROBIN and SKYLARK studies—demonstrated a significant clinical response within the first 3 days of treatment, with effects maintained for up to 45 days after treatment discontinuation.
In Phase III clinical trials, a meaningful clinical response was observed within 12–24 hours following a 60-hour infusion, with sustained effects lasting up to 30 days. However, due to potential adverse effects such as sedation, confusion, and in rare cases, syncope, brexanolone treatment is restricted to certified healthcare facilities under the FDA’s Risk Evaluation and Mitigation Strategy (REMS) program.
Zuranolone: Rapid Onset with Oral Administration
Zuranolone became the first FDAapproved oral neurosteroid for PPD in 2023. It is administered as a oncedaily 50 mg dose over a 14-day course and acts by modulating GABA-A receptors. By enhancing the effect of GABA, zuranolone exerts rapid anxiolytic and moodstabilizing effects within a few days. Since its mechanism does not directly involve the serotonin or dopamine systems, it presents a lower risk of drug interactions and a shorter duration of action.
A Non-Monoaminergic Pathway: New Antidepressants Targeting GABA
As an alternative to the slow-acting, serotonin-based mechanisms of traditional antidepressants, neurosteroids are opening a completely new therapeutic door: the GABA-A receptor system. Following childbirth, endogenous allopregnanolone levels drop, resulting in a form of "silencing" within the GABA-A system. This disruption is increasingly recognized as a key contributor to prominent postpartum depression symptoms such as anxiety, irritability, and insomnia. By positively modulating GABA-A receptors, zuranolone and brexanolone restore GABAergic tone and alleviate symptoms.
Emerging Oral Molecules: LYT-300 and LPCN-1154
LYT-300: This is a lipid-conjugated form of allopregnanolone. Phase I studies have shown that when administered orally, LYT-300 can achieve plasma concentrations comparable to IV brexanolone. A significant reduction in cortisol response to stress has also been reported. Phase II trials are currently being planned.
LPCN-1154: This is an oral formulation of brexanolone. According to 2024 data from Lipocine, it has demonstrated bioequivalent pharmacokinetics to IV brexanolone. Notably, no sedation has been observed. The compound is intended to be administered over a short 2-day treatment protocol.
A NEW GENERATION APPROACH IN THE TREATMENT OF SCHIZOPHRENIA
NBI-1117568

Numerous receptor systems are thought to be involved in the development and treatment of schizophrenia, including dopamine (especially D2, D3, D4), glutamate (NMDA), acetylcholine (M1 and M4 muscarinic), serotonin (5-HT2A), nicotinic, TAAR1 (Trace AmineAssociated Receptor 1), toll-like receptors (TLR) and cannabinoid CB2. This multilayered neurobiology not only adds to the complexity of the symptoms, but also to the difficulties in treatment.
The clinical journey that started with typical antipsychotics and then continued with atypical antipsychotics has led to significant gains in the treatment of schizophrenia, especially on positive symptoms. However, extrapyramidal side effects such as weight gain, metabolic disorders and inadequate effects on negative/cognitive symptoms brought about by these drugs have raised the same question in the minds of researchers: Is a treatment beyond the dopamine D2 receptor possible? For these reasons, efforts to identify alternative targets with a more favorable side effect profile have increased in recent years, and the cholinergic system, particularly muscarinic receptor subtypes, has become an area of interest in research. It has been demonstrated that M1 and M4 subtype muscarinic receptors may play a role in the pathophysiology of schizophrenia through their interactions with the dopaminergic system. In this context, the M1/M4 receptor agonist xanomelin, was formulated in combination with with trospium to reduce peripheral muscarinic side effects. This compound was approved by the US Food and Drug Administration (FDA) for the treatment of schizophrenia on September 26, 2024 . The increasing evidence that M4 receptors may provide therapeutic effects with fewer peripheral side effects compared to M1 receptors has led researchers to focus on this receptor. Preclinical studies have shown that regulation via M4 can suppress positive symptoms without directly blocking the D2 receptor or causing extrapyramidal side effects. The M4 receptor, which carries a lower risk of convulsions compared to M1 and has more limited peripheral effects, has attracted growing interest from researchers in recent years as a promising target for schizophrenia treatment. On August 28, 2024, Neurocrine Biosciences—a biopharmaceutical company headquartered in San Diego, California—announced the results of a Phase 2 clinical trial of NBI-1117568, an M4-selective agonist developed for the treatment of schizophrenia (3). This Phase 2 study was conducted with 210 adult participants with schizophrenia in an acute exacerbation or relapse phase and required hospitalization and aimed to evaluate the efficacy, safety, tolerability, and pharmacokinetic properties of NBI-1117568 in patients with schizophrenia. Participants who completed informed consent, discontinued their current antipsychotic treatment prior to study initiation, and were able to adhere to the protocol during the inpatient observation period were included in the study. Individuals with unstable medical conditions, a history of moderate to severe substance use disorder in the past, those at risk of suicide, or those deemed by the investigator to be unlikely to adhere to the protocol were excluded. The study, designed as a multicenter, randomized, double-blind, placebo-controlled trial, evaluated different dose regimens.
Muscarinic receptors are classified into five subtypes: M1 through M5. Of these, the M4 subtype is particularly notable for its ability to physiologically regulate dopamine release.
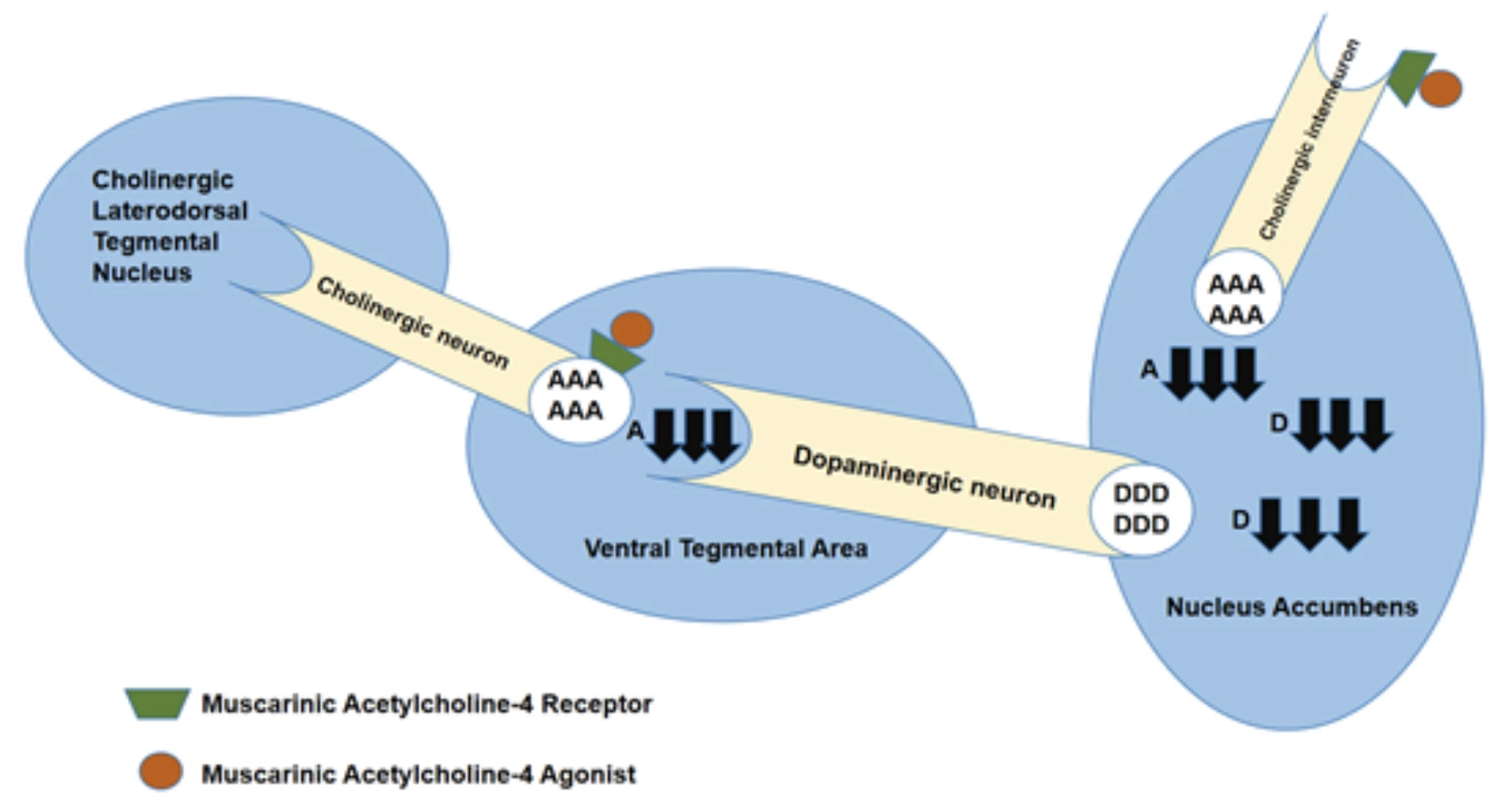
Acetylcholine (A) is released from cholinergic neurons located in the laterodorsal tegmental nucleus (LDT). Muscarinic acetylcholine receptor type 4 (M4) agonism activates autoreceptors on these cholinergic neurons. Activation of these receptors leads to a reduction in acetylcholine release. Consequently, dopamine neurons in the ventral tegmental area (VTA), which are normally stimulated by acetylcholine, become less active. This results in decreased dopamine release in the nucleus accumbens and a reduction in psychotic symptoms. Simultaneously, M4 agonism also activates autoreceptors on cholinergic interneurons located in the nucleus accumbens. This further reduces acetylcholine release in this region. The decline in acetylcholine levels in the nucleus accumbens leads to decreased activity of dopaminergic neurons projecting to this area. As a result, dopamine release in the nucleus accumbens is reduced, contributing to a decrease in psychotic symptoms.
Effect of NBI-1117568 on PANSS Total Score:
The table below summarizes the mean changes in PANSS total score and statistical significance levels for the dose regimens evaluated in the study..
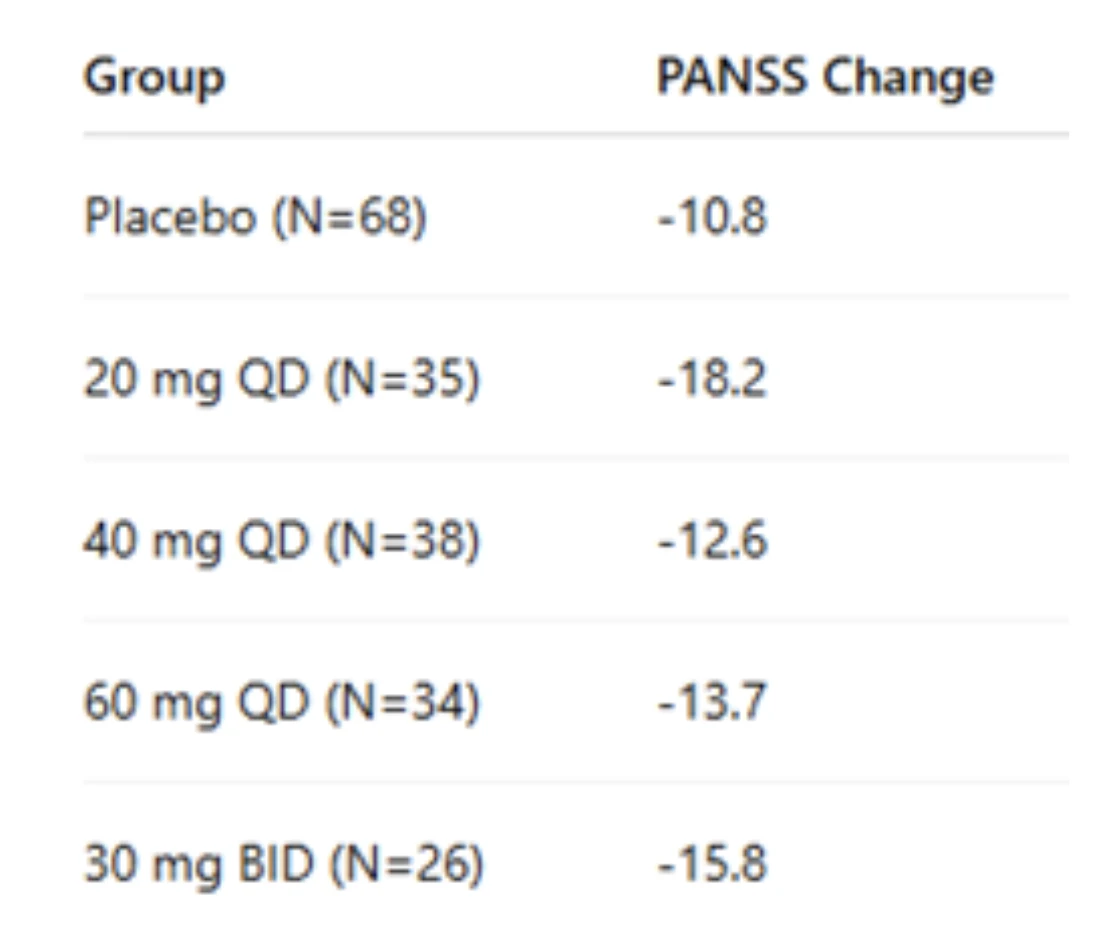
Among the groups administered a single daily dose of 20 mg, 40 mg, 60 mg, and two daily doses of 30 mg (total 60 mg/day) of NBI-1117568, the most significant clinical improvement was observed at week 6 in the 20 mg/day dose group; a reduction of 18.2 points in the PANSS total score was achieved in this group. This improvement was found to be statistically significant compared to placebo (p=0.011, effect size: 0.61).
Safety and Tolerability of NBI-1117568
In terms of safety profile, NBI-1117568 was generally well tolerated at all dose levels. The rates of discontinuation due to treatmentrelated adverse events were similar between the drug-treated groups and the placebo group. The most commonly reported side effects included drowsiness, dizziness, and headache. Gastrointestinal side effects such as nausea and constipation were observed at a low frequency and were similar to the placebo group. Additionally, the incidence of cardiovascular events was low and not clinically significant. Weight gain did not increase in the NBI-1117568 group compared to the placebo. Furthermore, drug-related extrapyramidal symptoms were rarely reported. However, the lack of reporting of PANSS scale scores and the absence of statistically significant improvement in PANSS total scores at doses higher than 20 mg/day are notable limitations of the study. The study was conducted in a single center, which may further limit the generalizability of the results.
Despite its limitations, NBI-1117568's innovative mechanism of action and promising early findings are generating excitement that it may herald a new era in schizophrenia treatment.
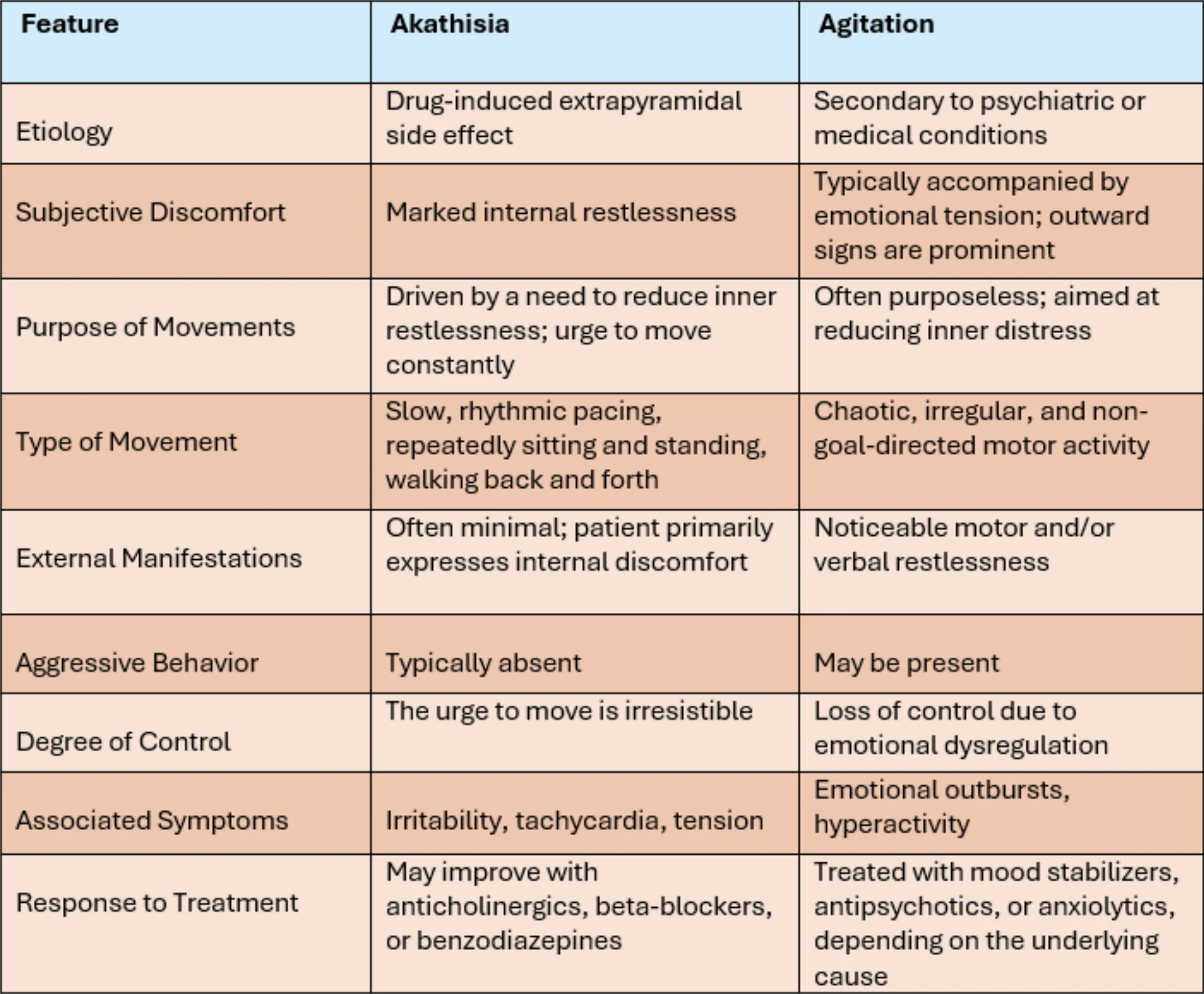
Akathisia or Agitation?
Akathisia and agitation are two clinically significant and distinct conditions that are often mistaken for one another. A thorough assessment of the patient's complaints and direct clinical observation is critical to making an accurate diagnosis. The purpose and nature of the movements as well as accompanying emotional symptoms serve as important diagnostic clues. Misidentification can lead to inappropriate pharmacological interventions, potentially exacerbating the patient’s clinical picture.
Akathisia
Akathisia typically arises as an extrapyramidal side effect of medications and is characterized by marked internal restlessness. Patients often engage in repetitive, rhythmic movements—such as pacing or repeatedly sitting and standing—in an attempt to alleviate this discomfort. Despite these observable behaviors, external manifestations are usually limited; patients primarily report internal distress, and aggressive behavior is uncommon. Treatment may include anticholinergics, beta- blockers, or benzodiazepines.
Agitation
Agitation, on the other hand, is often secondary to underlying psychiatric or medical conditions and is characterized by pronounced motor or verbal restlessness accompanied by emotional tension. The movements tend to be chaotic, disorganized, and generally lack a clear purpose. Unlike akathisia, agitation may involve aggressive behavior. Management should be tailored to the underlying cause and may include mood stabilizers, antipsychotics, or anxiolytics.
M.D. Ayşe Bağcı
References:
DOI:10.1136/bmjopen-2017-017406
DOI:10.1192/j.eurpsy.2021.1287
DOI:10.1097/yco.0b013e32832a16da
DOI:10.3109/15622975.2015.1132007
DOI:10.1017/neu.2022.9
A NEW MOLECULE:
LURASIDONE IN CHILDREN AND ADOLESCENTS

The limited availability of approved pharmacological treatments in child psychiatry complicates treatment in cases with complex features and highlights the growing importance of transdiagnostic approaches.
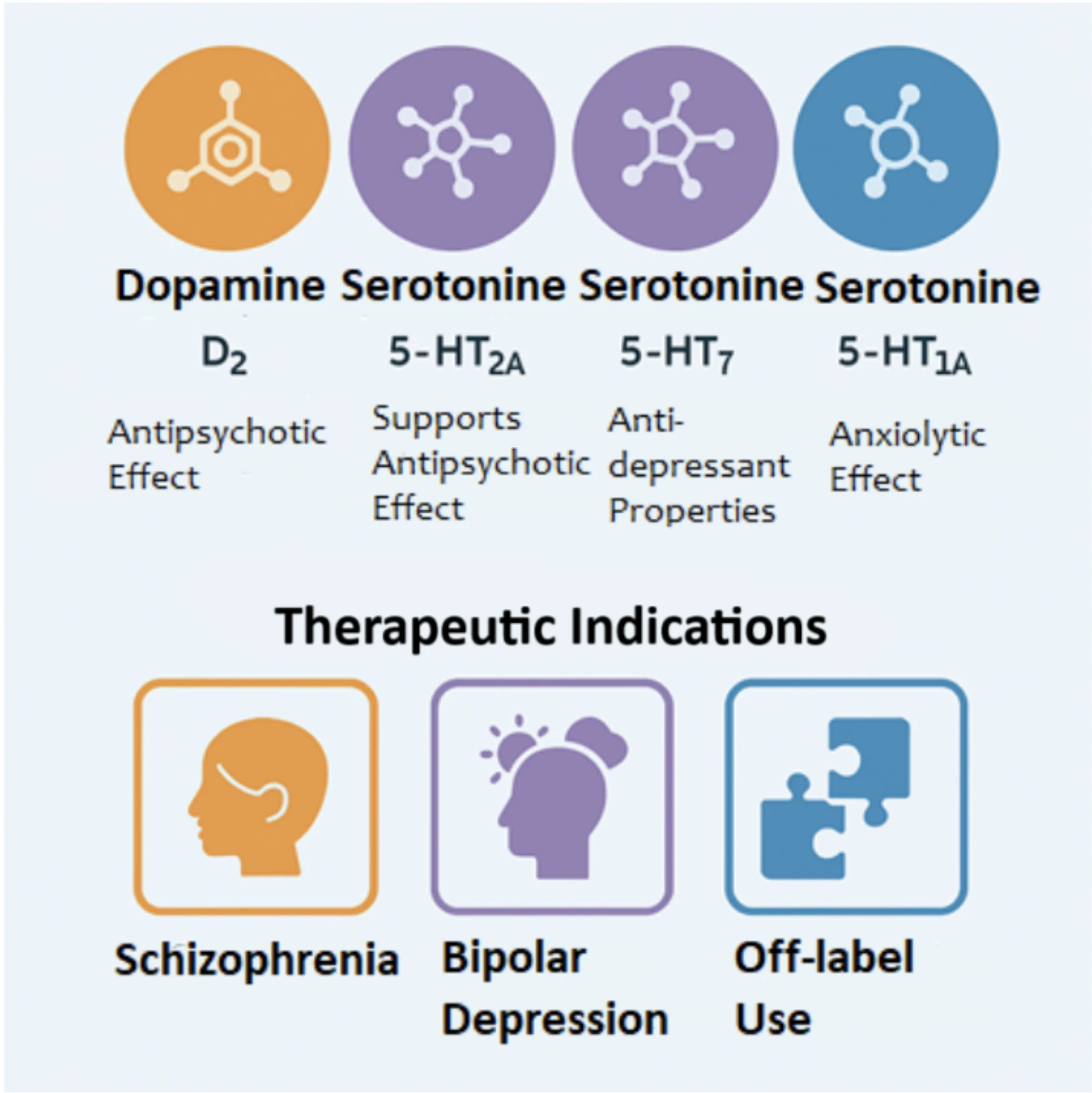
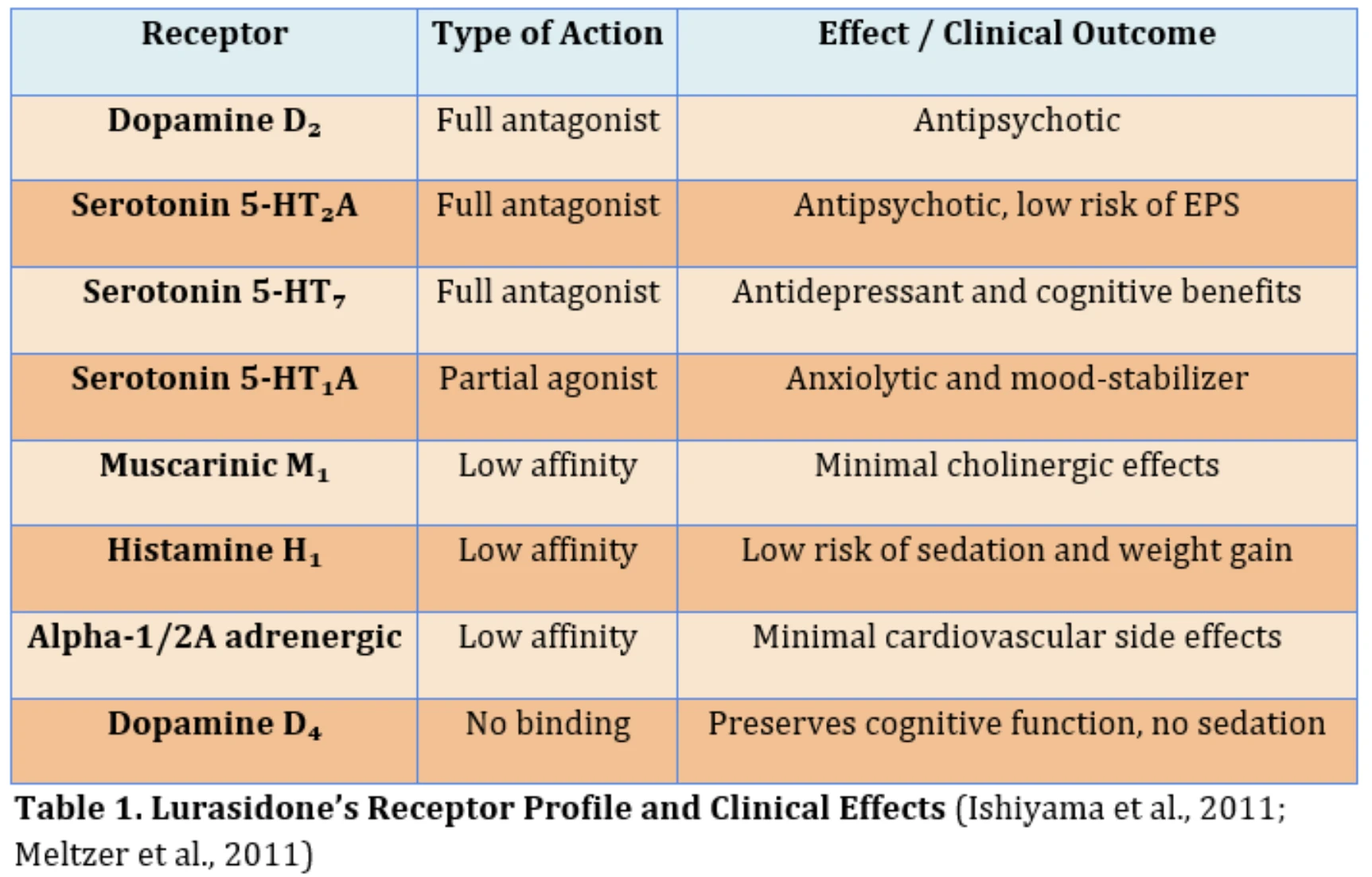
Lurasidone is a novel atypical antipsychotic that stands out due to its unique pharmacodynamic profile on serotonin and dopamine systems. Lurasidone was approved in 2017 for the treatment of early-onset schizophrenia (EOS) in individuals aged 13 and above, and in 2018 for pediatric bipolar depression in those aged 10 and above.
The IACAPAP recommend lurasidone as a first-line treatment for pediatric bipolar depression, alongside the olanzapinefluoxetine combination. Lurasidone also offers antidepressant effects through its profile. Its mechanism is summarized in Table 1.
Pharmacokinetics and Pharmacodynamics:
Lurasidone is metabolized via the CYP3A4 isoenzyme and is contraindicated with strong inhibitors or inducers of this enzyme (Azhar & Shaban, 2023). Its bioavailability increases when taken with food. In children and adolescents, lurasidone is shown to be generally well tolerated. The mostcommon side effects include sedation, increased appetite, and nausea. More serious adverse effects such as weight gain, QTc prolongation, elevated prolactin levels, and extrapyramidal symptoms are observed at very low rates (Findling et al., 2015; Correll et al., 2015). Lurasidone’s low sedative effects and lack of negative impact on cognitive function provide clinical advantages in maintaining academic and social functioning (DelBello et al., 2017). Lurasidone shows a doseproportional and predictable pharmacokinetic profile in children and adolescents. Daily doses of 20–80 mg are generally well tolerated, whereas doses above 120 mg carry an increased risk of adverse effects (Findling et al., 2015).
Efficacy Findings: Early-Onset Schizophrenia (EOS)
In EOS treatment, lurasidone stands out as an effective, safe, and metabolically advantageous antipsychotic.
A randomized placebo-controlled study by Goldman et al. (2017) reported that lurasidone significantly improved PANSS and CGI-S scores compared to placebo. A network meta-analysis by Arango et al. (2020) showed that lurasidone improved these scores with less weight gain than agents like olanzapine and risperidone In a twoyear open-label extension study, Correll et al. (2022) reported a 58.3% reduction in symptoms, reflected in decreased PANSS total scores. Remission and recovery rates reached approximately 50% by the second year, with a low adverse event profile. These findings support the long-term efficacy of lurasidone in adolescent schizophrenia.
Efficacy Findings: Bipolar Disorder
Lurasidone has proven to be an effective and safe treatment option for adolescent bipolar depression in both short- and long-term use. Studies by DelBello et al. (2017, 2021) demonstrated a sustained reduction in depressive symptoms over two years, with mostly mild to moderate side effects.
High response and remission rates were accompanied by marked improvements in functional outcomes and quality of life. Its low metabolic risk particularly offers an advantage in long-term pediatric treatment.
Diao et al. (2022) found lurasidone to be superior to quetiapine in terms of cognitive performance. With its serotonergic effects, BDNF enhancement, and antioxidant pathways, lurasidone provides comprehensive benefits for both mood regulation and cognitive function.
Bridge symptoms such as insomnia and irritability may lead to transitions between depressive and manic states, especially in adolescents with mixed features. Studies by Singh et al. (2020, 2023) showed that lurasidone was effective in managing cases with these bridge symptoms, even when subsyndromal hypomanic symptoms were present, indicating improvement across both symptom clusters. No increased risk of manic switch was observed and the remission rates were significant. As both studies were based on the same randomized trial, results should be considered as hypothesis-generating.
Off-label Use:
Case series have reported that lurasidone can help with irritability, aggression, and impulsivity in conditions like autism spectrum disorder (ASD) and Tourette syndrome. Improvements in home behavior and social functioning have been noted with adjunctive treatment with lurasidone (Colizzi et al., 2021; Gupta & Hoover, 2020; Millard & McLaren, 2014). However, these findings are limited based on small samples, combined treatments, and uncontrolled study designs.
Conclusion:
Lurasidone shows promise as an effective and well-tolerated treatment option in child and adolescent psychiatry. However, diagnostic challenges in this age group must be considered. Adolescent psychopathology often presents atypically and fluctuates, with frequent transitions between diagnoses—supporting the need for symptom-based treatment approaches. Moreover, children's limited ability to express themselves, reduced reliability of anamnesis, and stigma-related delays in diagnosis can postpone treatment initiation.
In conclusion, lurasidone is an antipsychotic agent that offers symptomatic relief in early-onset schizophrenia and bipolar depression with metabolic advantages and cognitive-sparing properties. Nevertheless, further long-term and transdiagnostic studies are needed, considering the heterogeneity of psychopathology in youth. Controlled clinical trials are expected to clarify lurasidone’s potential indications and its use across overlapping symptom domains.
It has been shown that cases with bridge symptoms respond to lurasidone, and although subsyndromal hypomanic features may be present, lurasidone has provided improvement in both symptom clusters.
- DOI: 10.1007/s00787-019-01425-2
- DOI: 10.1016/j.tice.2025.1473457
- DOI: 10.1016/j.psychres.2020.113609
- DOI: 10.1002/wps.20204
- DOI: 10.1016/j.jaac.2017.10.006
- DOI: 10.1089/cap.2021.0040
- DOI: 10.3390/ph15111403
- DOI: 10.1089/cap.2014.0105
- DOI: 10.1089/cap.2016.0189
- DOI: 10.1089/cap.2019.0069
- DOI: 10.1254/jphs.11082FP
- DOI: 10.1007/s10803-014-2304-3
- DOI: 10.1176/appi.ajp.2011.10060908
- DOI: 10.1089/cap.2013.0116
- DOI: 10.1177/0897190021997011
- DOI: 10.1089/cap.2020.0018
- DOI: 10.2174/1570159X206667112625