
TAP Psychopharmacology Bulletin - New Horizons / 2026 February
Prepared for clinicians and researchers working in the field of psychiatry.
Bulletin Subscription
Be the first to know when new issues are published! Apply now for free subscription.
Free • Cancel anytime you want
News
Lumateperone Approved as Adjunctive Therapy for Major Depressive Disorder
Lumateperone has shown promising results in schizophrenia and bipolar disorder due to its modulatory effects on serotonin, dopamine, and glutamate neuronal transmission. These findings have encouraged the investigation of the drug's potential effects on Major Depressive Disorder (MDD). The study, included 480 participants aged 18-65 with moderate to severe depression. Participants were randomized to receive either lumateperone plus existing antidepressant therapy or placebo plus existing therapy for six weeks. Lumateperone as an adjunctive treatment provided robust and clinically significant benefits. On Day 43, a significant and clinically meaningful reduction in Montgomery-Åsberg Depression Rating Scale (MADRS) scores, significant improvement in Clinical Global Impression–Severity (CGI-S) scores, and improvement in patient- reported depression levels (QIDSR-16) were observed compared to the placebo group.
- A New Phase in Alzheimer’s Disease Treatment
- Current Debates on Autism
- Pimavanserin
- Guanfacine and Prefrontal Cortical Balance
- Phosphatidyiserine and Citicoline
- Integrity of Blinding in Psychiatric Randomized Controlled Trials
Treatment was generally well-tolerated in the lumateperone-added group. No significant differences were detected between the two groups regarding metabolic parameters, weight change, changes in prolactin levels, extrapyramidal symptoms, or suicidality. Consequently, these findings support that lumateperone 42 mg, when used as an adjunct to antidepressant treatment, provides statistically and clinically significant improvement in depressive symptoms and disease severity in MDD patients with an inadequate response to therapy. On November 6, 2025, lumateperone received FDA approval for use as an adjunctive therapy to an antidepressant for MDD. Studies evaluating its long-term efficacy are ongoing.
Uzm. Dr. Elif Özge Aktaş-Keskin
DOI: 10.1176/appi.ajp.20250292
Does Prenatal Exposure to Benzodiazepines and Z-drugs Increase the Risk of Neurodevelopmental Disorders?
Benzodiazepines and Z-drugs are commonly used to treat anxiety and insomnia in women of childbearing age. However, concerns persist regarding potential effects due to the critical role of GABAergic signaling in fetal brain development. A study published in The British Journal of Psychiatry investigated whether prenatal exposure to benzodiazepine receptor agonists (BZRAs) is associated with long-term neurodevelopmental disorders (LNDDs) in children. Using the South Korean National Health Insurance Service database, the study examined 1.55 million births occurring between 2011 and 2014, with children followed until 2023.
Intrauterine BZRA exposure was identified in a total of 5,967 children. BZRA exposure was associated with a 25% increase in the risk of LNDDs. Analyses of the entire cohort showed that the risk increased with longer durations of exposure. Benzodiazepine use alone was found to increase the risk more significantly, while the effect of Z-drugs was weaker. Although trimester-based models showed increased risk across all trimesters, exposure during the second and third trimesters was associated with the most pronounced risk.
These findings suggest that prenatal BZRA exposure may be linked to long-term neurodevelopmental risks in children. Highlighting the potential disruption of GABA-mediated neurodevelopment, the authors recommend caution in prescribing during pregnancy, prioritizing non- pharmacological alternatives, and keeping treatment duration as short as possible when medication is necessary.
Uzm. Dr. Elif Özge Aktaş-Keskin
DOI: 10.1192/bjp.2025.10481
Metformin: Antipsychotic Side Effect Rescuer and Treatment Adherence Enhancer
Weight gain is a common, difficult- to-prevent, and comorbidity- increasing side effect of antipsychotic medications.
A systematic review has shown that metformin is the only agent that has an effect on weight gain in both the short-term (less than 12 weeks) and long-term. Metformin has been shown to have consistent weight-gain- preventing effects, particularlywhen added to the treatment of patients using olanzapine, who are at high risk for metabolic syndrome.
Similarly, the effect of metformin on hyperprolactinemia, a side effect of olanzapine, has been investigated, and significant reductions in prolactin levels were observed after eight weeks of metformin intervention.
According to a follow-up study in a UK country cohort, after taking Second Generation Antipsychotics (SGAs), the mean weight change was +4.16% in the SGA-only group, while it was -0.65% in the SGA plus metformin group. In patients without diabetes who were receiving SGA, it was observed that when metformin was initiated, adherence to the medication increased and the duration of treatment was extended.
In conclusion, the addition of metformin to antipsychotic treatment appears promising due to its ability to reduce side effects and improve treatment adherence.
Dr. Furkan Yazıcı
DOI: 10.1002/npr2.70061
- Prof. Dr. M. Kemal SAYAR
- Prof. Dr. Mesut ÇETİN
- Prof. Dr. Feyza ARICIOĞLU
- Prof. Dr. Ali Saffet Gönül
- M.D. Gözde Çolak
- Yiğit Erdoğan
Its content is independent of the pharmaceutical industry.
Editorial...

Dear Colleagues,
We have had an autumn marked by considerable activity and new developments in the field of psychopharmacology. The inclusion of antipsychotics such as brexpiprazole and cariprazine in reimbursement coverage has broadened access, enabling their use across a much wider patient population. Internationally, debate continues over the clinical impact of newly developed Alzheimer’s therapies, while the potential place of psychedelics in treatment is still being actively investigated. While we were experiencing these exciting developments, we were shaken by sad news in December. We lost Prof. Dan Stein, a leading researcher in anxiety disorders. A close friend of the Psychopharmacology Society and a valued speaker at our congresses, Prof. Stein is commemorated in this issue in an article that aims to introduce him especially to young researchers.
Even as new agents enter routine clinical practice, longstanding challenges persist Medication-related weight gain is among the most prominent of these problems. Obesity contributes to a wide range of comorbidities. It has long been recognized that individuals with bipolar disorder and schizophrenia carry elevated cardiovascular risk independent of pharmacotherapy; accordingly, people living with psychotic disorders have a life expectancy that is approximately 20 years shorter than that of the general population. In depression, particularly when the illness follows a recurrent course, the risk of cardiovascular and endocrine disease is also increased.
Many psychotropic medications, through H1 and 5-HT2C antagonism, can increase appetite and also adversely affect metabolic function. Alongside agents such as clozapine—often used in treatment-resistant cases—prolonged olanzapine treatment (notably, notably, it is among the least frequently discontinued antipsychotics) and very long-acting formulations such as paliperidone can lead to weight gain. Notably, weight gain may occur even with medications regarded as relatively weight-neutral. In the context of polypharmacy, weight gain often becomes difficult to avoid.
In addition to behavioral strategies, pharmacologic approaches are also frequently considered. Among these, metformin is the most commonly used medication. Initiating metformin early may be more effective in preventing weight gain. A recent meta-analysis reported that metformin is effective in reducing antipsychotic-associated weight gain, with a mean weight change of −3.32 kg, and also demonstrated a meaningful reduction in body mass index.
While the evidence base is largely limited to trials of around 12 weeks, we do not have strong evidence regarding long-term metformin use; however, given its favorable safety profile, longer-term use is not generally considered problematic in clinical practice. Because vitamin B12 and thiamine deficiencies are among the most common long-term adverse effects, periodic monitoring of B12 and thiamine levels is advisable. Lactic acidosis and other serious adverse events (such as pancreatitis) are exceedingly rare, but may occur in patients with advanced renal, hepatic, or respiratory failure, sepsis, or heavy alcohol use. No meaningful difference in efficacy has been demonstrated between higher doses (>1000 mg) and lower doses (<1000 mg). Although GLP-1 receptor agonists can produce substantial weight loss in obesity, a large matched cohort study including more than 160,000 individuals reported a higher incidence of new-onset psychiatric diagnoses among users of liraglutide and semaglutide compared with individuals not treated with GLP-1 receptor agonists The risk was approximately doubled for any psychiatric disorder, major depression, anxiety disorders, and suicidal behavior. These findings underscore the importance of taking a detailed psychiatric history before initiation, ensuring close clinical follow-up during treatment, and conducting prospective studies to clarify causality. For patients with psychiatric illness, careful pre-treatment assessment and ongoing monitoring should therefore be regarded as essential. Evidence on the use of GLP-1 receptor agonists in psychiatric populations continues to emerge. Especially as oral formulations gain approval, it would be prudent to keep these findings in mind until clearer consensus becomes available.
Wishing you health and peace
in the years ahead,

A NEW PHASE IN ALZHEIMER’S DISEASE TREATMENT:
CURRENT THERAPEUTIC APPROACHES AND CLINICAL ADVANCES
Spc. Dr. Duygu Keskin Gökçelli

Alzheimer’s disease (AD) is a progressive neurodegenerative disorder that continues to represent the most prevalent cause of dementia worldwide. Clinically, it is progressive impairments in memory, learning, attention, executive functions, and activities of daily living. Although it typically manifests after the age of 65, early-onset forms have also been reported.
Pathophysiology and Current Treatments
Pathologically, AD is marked by extracellular accumulation of amyloid-β (particularly Aβ42) forming senile plaques, and intracellular hyperphosphorylation of tau proteins leading to neurofibrillary tangles, accompanied by significant neuronal loss and synaptic degeneration. These alterations predominantly cause atrophy in the hippocampus and temporal lobes. Accumulation of amyloid-β peptides is considered central to the disease, initiating synaptic dysfunction and neurodegeneration. Tau protein plays a key role in stabilizing microtubules and preserving neuronal structure under physiological conditions; however, when hyperphosphorylated, it dissociates from microtubules and aggregates into neurofibrillary tangles, ultimately resulting in substantial neuronal damage. Especially phosphorylated tau (p-τ) forms have been closely associated with neuronal loss. Recent studies suggest that amyloid-β and tau pathologies interact rather than act independently, jointly contributing to synaptic failure and neuronal death. Furthermore, cholinergic system dysfunction and glutamate-mediated excitotoxicity accelerate cognitive decline, while microglial activation and chronic neuroinflammation contribute to disease pathogenesis.
Until 2021, therapeutic options were largely limited to symptomatic agents. Acetylcholinesterase inhibitors (donepezil, rivastigmine, galantamine) are recommended for mild to moderate stages, while the NMDA receptor antagonist memantine is used alone or in combination therapy for moderate to severe stages.
Multiple therapeutic approaches for Alzheimer’s disease have emerged in recent years. Amyloid-β–targeted monoclonal antibodies represents a key advancement.
Behavioral and affective symptoms are managed with antidepressants, antipsychotics, and supportive interventions such as exercise, cognitive rehabilitation, balanced nutrition, environmental modifications, and caregiver support.
Current Therapeutic Approaches
For the AH therapy, Aducanumab and lecanemab have received FDA approval and demonstrated reductions in amyloid plaque burden, and donanemab followed with FDA approval in 2024. However, the clinical benefits of these agents remain limited, and concerns regarding their safety profiles continue to be discussed. Tau- targeted therapies, including immunotherapy-based anti-tau antibodies and vaccine approaches, are currently under clinical investigation. Immunotherapy and vaccine-based approaches aim to stimulate the immune system to recognize and clear pathological tau proteins, thereby reducing tau aggregation and intercellular spread. Strategies targeting tau phosphorylation seek to inhibit the enzymes responsible for abnormal phosphorylation, preventing neurofibrillary tangle formation and neuronal damage.
Another promising direction in Alzheimer’s treatment is drug repurposing. In this context, the tyrosine kinase inhibitor nilotinib, originally developed for chronic myeloid leukemia, has been proposed to reduce amyloid plaque and hyperphosphorylated tau accumulation and to help limit hippocampal volume loss.
Advances in understanding the role of neuroinflammation in the pathogenesis of Alzheimer’s disease have stimulated interest in developing new therapeutic strategies. These strategies include TNF-α inhibitors, approaches that modulate microglial activity, and interventions targeting the gut microbiota. Additionally, neuroprotective agents such as NMDA receptor modulators, stem cell therapies, and growth factors are being investigated for their potential to preserve synaptic plasticity and limit neuronal injury.
Non-pharmacological interventions also play an important role in treatment. Neuromodulation techniques such as repetitive transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS), and transcranial pulse stimulation (TPS) are currently under investigation and are often complemented by lifestyle interventions including physical activity, dietary modification, and cognitive training. Additionally, the therapeutic potential of agents such as lithium, metformin, levetiracetam, and sodium benzoate is also being investigated. Finally, to enhance targeted drug delivery, advanced delivery systems are being developed. These advances show a clear shift in Alzheimer’s treatment from symptom-focused care toward a more innovative, multidisciplinary approach that directly addresses the disease’s underlying pathology.
Conclusion
Alzheimer’s disease remains a complex and multifactorial neurodegenerative disorder for which a definitive cure has not yet been achieved. Recent advances in disease- modifying therapies targeting amyloid-β, tau, neuroinflammation, and neuroprotective mechanisms are gradually approaching broader clinical use and offer the potential to influence disease progression rather than only relieve symptoms. However, long-term data on efficacy and safety are still required. Moving forward, the integration of pharmacological, biological, and non-pharmacological strategies is expected to support more comprehensive and personalized treatment approaches, marking a promising new phase in the management of Alzheimer’s disease.
- DOI: 10.1038/s41380-021-01249-0
- DOI: 10.3390/ijms232112841
- DOI: 10.1038/s41593-020-0687-6
- DOI: 10.1056/NEJMoa2212948
- DOI: 10.1007/s40265-024-02087-4
- DOI: 10.1038/s41582-023-00883-2
- DOI: 10.1002/ana.25775
- DOI: 10.3233/ADR-220097
- DOI: 10.4103/1673-5374.380874
- DOI: 10.1093/brain/awae292
- DOI: 10.3390/ijms22158208
- DOI: 10.3389/fphar.2022.979682
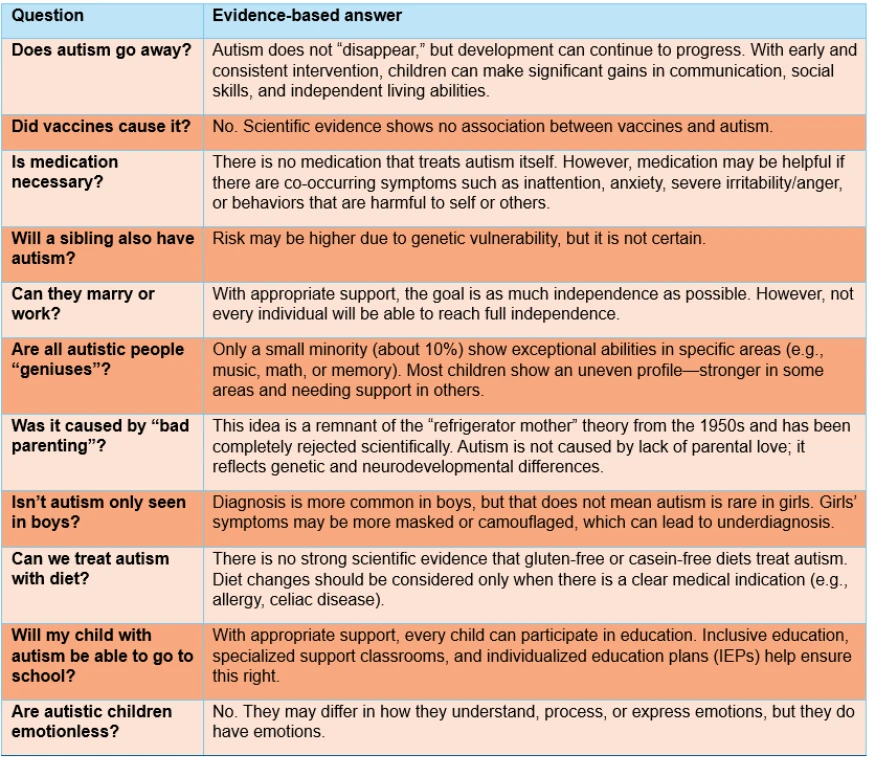
CURRENT DEBATES ON AUTISM:
LATEST PAPERS AND COMMENTS FROM SPECIALISTS
Dr. Ece Pabuşçu

In 1943 and 1944, Leo Kanner and Hans Asperger, reportedly unaware of each other’s work, independently published papers that for the first time defined autism as a disorder distinct from schizophrenia. Since then, due to factors such as increased recognition of autism, widespread screening programs, improved access to healthcare, changes in diagnostic criteria, and exposure to environmental toxins, this disorder, once thought to occur in about 1 in 10,000 individuals, has now been identified in approximately 3% of the population.
The approximately 277-fold increase in prevalence over the past 60 years, together with the continued lack of a definitive curative solution, has led scientists to focus on etiological factors with the aim of preventing autism from occurring in the first place.
Vaccines and Autism Claims
Andrew Wakefield and his colleagues’ 1998 article published in The Lancet, which controversially implicated the Measles, Mumps, and Rubella (MMR) vaccine as a direct cause of autism, ignited a fierce and enduring global debate regarding the etiology of autism spectrum disorders. This highly sensationalized publication not only fueled a significant and dangerous rise in anti- vaccine sentiment across the world but also paved the way for the widespread dissemination of unscientific notions and conspiracy theories, despite the fact that the study contained serious methodological flaws, small sample sizes, and profound ethical violations.
The paper’s manipulative data selection and lack of scientific rigor eventually led The Lancet to retract the article entirely in 2010, fully twelve years after its initial publication. Furthermore, the severity of the misconduct resulted in the revocation of Wakefield’s medical license by the General Medical Council in the United Kingdom. Following this retraction, the scientific community responded with robust meta-analyses and extensive large-scale cohort studies involving millions of children, all of which have consistently failed to find any causal link between vaccines or components and the development of autism.
Nevertheless, the profound, life- altering, and indefinite impact of an autism diagnosis on families often drives parents to search desperately for a concrete cause or someone to blame for their child's condition. Consequently, even in the face of overwhelming and incontrovertible scientific evidence to the contrary, the scapegoating of vaccines persists at a societal level, sustained by fear, misinformation, and the human desire for a simple explanation to a complex biological condition.
Acetaminophen Use During Pregnancy
While research on the etiology of autism continues, a new review was published in September 2025 examining the relationship between acetaminophen use during pregnancy and autism. Because of methodological heterogeneity, no meta-analysis could be conducted; instead, the Navigation Guide methodology was applied. The review examined 46 publications, eight of which (six original) evaluated the acetaminophen–autism association. The results suggested a possible link, but the authors clearly emphasized that methodological limitations and biases prevent any conclusion of causality.
In this context, the factor of “confounding by indication” is particularly important. Pregnant women do not use acetaminophen randomly; it is typically taken for medical reasons such as fever, inflammation, infection, or autoimmune processes— conditions that themselves may increase the prenatal risk of autism. Therefore, assessing the acetaminophen–autism relationship in isolation can lead to misleading interpretations. Twin studies and GWAS findings indicate that 70–90% of autism risk is attributable to genetic factors.
Environmental influences shape this risk through interaction with genetic susceptibility. It is crucial to distinguish causation from correlation: the simultaneous occurrence of two variables does not prove that one causes the other. For example, both ice cream sales and shark attacks increase during the summer months, but this does not mean that ice cream consumption causes shark attacks. It is evident that science must continue to make progress in understanding the causes and treatment of autism.
Pregnant women do not use acetaminophen randomly; it is typically taken for medical reasons such as fever, inflammation, infection, or autoimmune processes—conditions that themselves may increase the prenatal risk of autism.
Expanding prospective longitudinal studies, investigating gene–environment interactions, and developing biomarker research will strengthen the knowledge base in this field. Each claim not grounded in scientific evidence, however, risks placing additional blame on mothers of autistic children. Referring to acetaminophen—used as an antipyretic during pregnancy and long regarded as the relatively safe option— has placed yet another burden on mothers of autistic children.
Conclusion
As research on autism continues, efforts aimed at preventing stigmatization, disseminating accurate information, facilitating access to special education, and improving the quality of life of individuals with autism and their families should not be overlooked.
In this context, developing a risk- communication strategy that protects both maternal health and fetal development while also strengthening public trust should be one of the most important goals in the period ahead.
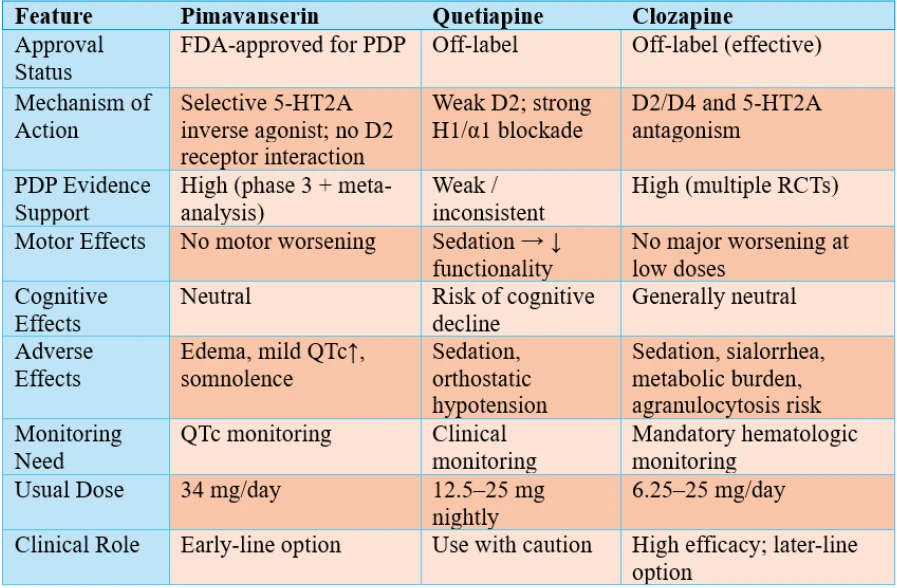
PIMAVANSERIN:
CURRENT EVIDENCE AND CLINICAL POSITIONING
Dr. Özge Bıldırcın

Pimavanserin originally developed as a selective 5-HT2A receptor inverse agonist/antagonist without dopamine D2 receptor interaction. Since its FDA approval, for the treatment of Parkinson’s disease psychosis (PDP), it has addressed an important unmet clinical need by providing antipsychotic efficacy without exacerbating motor symptoms. The ability to target psychotic symptoms while preserving dopaminergic function is the key differentiator of pimavanserin from traditional antipsychotics.
Evidence in Parkinson's Disease PsychosisThe most robust clinical data for pimavanserin come from the PDP population. In the pivotal phase 3 trial by Cummings et al., pimavanserin 40 mg demonstrated a significant reduction in positive psychotic symptoms measured by the SAPS-PD (Scale for the Assessment of Positive Symptoms in Parkinson's Disease), without meaningful deterioration in motor performance. Subsequent comparative analyses have consistently supported these findings.
In a recent network meta-analysis including 19 studies and 1242 patients, Yunusa et al. showed that pimavanserin and clozapine were the most effective agents in reducing psychotic symptoms in PDP. Pimavanserin was associated with significant clinical improvement in Clinical Global Impression-Severity scores and reductions in delusions.
- DOI: 10.1210/en.2013-1934
- DOI: 10.3389/fendo.2021.721135
- DOI: 10.1016/j.drudis.2016.01.013
- DOI: 10.1186/s12974-019-1638-6
- DOI: 10.1210/en.2011-1443
- DOI: 10.3389/fphar.2023.1063033
- DOI: 10.1523/JNEUROSCI.3262-11.2011
- DOI: 10.1111/dom.16453
- DOI: 10.1016/j.physbeh.2024.114565
- DOI: 10.1016/j.physbeh.2019.03.026
- DOI: 10.1038/s41386-018-0010-3
Although quetiapine did not worsen motor symptoms, its potential association with cognitive decline has contributed to a growing clinical preference for pimavanserin. It provides particular added value in patients who are motor-vulnerable, cognitively impaired, or at high risk if dopaminergic therapy is modified.
While clozapine may offer slightly superior efficacy, the absence of hematologic monitoring requirements and its more favorable tolerability profile position pimavanserin as an early-line therapeutic choice in PDP.
Findings regarding pimavanserin in dementia-related psychosis are more heterogeneous. In Alzheimer’s disease cohorts, efficacy appears modest and short-lived. However, the phase 3 HARMONY relapse- prevention trial, which included multiple dementia subtypes, demonstrated that continuation of pimavanserin reduced relapse risk by nearly two-thirds. Early termination of the study and the heterogeneous sample have limited the regulatory translation of these results.
As a result, pimavanserin currently lacks formal approval for dementia psychosis. Nevertheless, in settings such as Lewy body dementia—where both psychosis and parkinsonism frequently coexist. It may be considered off-label with careful risk–benefit assessment and cardiac monitoring.
Research in Schizophrenia and DepressionA theoretical advantage of pimavanserin in schizophrenia was its potential to modulate glutamatergic/ dopaminergic pathways without D2 blockade.
The phase 2 ADVANCE study found statistically significant but clinically modest improvements in Negative Symptom Assessment-16 (NSA-16) scores.
The confirmatory phase 3 trial failed to meet its primary outcomes, leading to discontinuation of the development program.Similarly, early encouraging results from the CLARITY phase 2 trial in treatment-resistant major depressive disorder were not replicated in subsequent phase 3 studies. Consequently, industry-sponsored development of pimavanserin outside the PDP indication has been halted.
Clinical Positioning and Practical Considerations
In contemporary practice, pimavanserin is increasingly recognized as a first-line therapy in PDP, particularly favored for its motor-sparing properties. It offers an alternative to the stepwise strategy of initiating quetiapine followed by clozapine if response is inadequate.
In dementia-related psychosis, a cautious approach is warranted. If off-label use is contemplated, clinicians should ensure thorough patient and caregiver education concerning expected benefits, QTc- prolongation risk, fall potential, and treatment costs. Once symptoms stabilize, abrupt discontinuation should be avoided to reduce relapse risk.
In schizophrenia and major depressive disorder, current evidence does not support routine clinical use. In such populations, pimavanserin should be limited to experimental or compassionate-use scenarios.
Overall Assessment and Future DirectionsPimavanserin represents an innovative pharmacological strategy. Its well-established efficacy and motor tolerability in PDP have already strengthened its role in neuropsychiatric practice. In dementia psychosis, its potential remains promising yet inconclusive; in schizophrenia and depression, its role appears limited. Notably, pimavanserin is generally well tolerated even in older and frail patients. Future research may better define its positioning. Based on current evidence, however, its principal role is firmly established in the management of Parkinson’s disease psychosis.
GUANFACINE AND PREFRONTAL CORTICAL BALANCE:
NEUROBIOLOGICAL MECHANISMS AND CLINICAL IMPLICATIONS OF Α2A-ADRENERGIC AGONISM
Dr. Gülenay Korkmaz Şahin

Impulsivity, inattention, hyperactivity, and conduct problems...
These symptoms do not merely define various psychiatric clinical presentations; they also point toward disturbances in the complex neurobiological equilibrium of the prefrontal cortex.
The prefrontal cortex (PFC) plays a central role in the coordination of attention, impulse control, emotional regulation, and executive functions. This region regulates imputs originating from subcortical structures through a "top-down" mechanism. This regulatory process enables the individual to exhibit more balanced, goal-oriented, and socially appropriate behaviors in response to environmental stimuli. Top-down regulation is facilitated via inhibitory projections extending from the prefrontal cortex to subcortical structures. This network relies on a delicate neurochemical balance maintained through noradrenergic α2A receptors and dopaminergic D1 receptors. Optimal levels of catecholaminergic activity improve the "signal-to-noise" ratio within neuronal networks; consequently, goal-directed neural signals are amplified while irrelevant or disruptive inputs are suppressed, rendering processes of attention, planning, and behavioral control more efficient.
The weakening of these regulatory circuits forms the neurobiological basis for the behavioral disinhibition and emotional lability observed particularly in Attention-Deficit/ Hyperactivity Disorder (ADHD), autism spectrum disorder, and impulse control disorders.
Guanfacine, as a selective α2A- adrenergic receptor agonist, activates postsynaptic α2A receptors located on the dendritic spines of pyramidal glutamatergic neurons in the prefrontal cortex. This activation leads to the inhibition of the Hyperpolarization-activated Cyclic Nucleotide-gated (HCN) channel signaling pathway (cAMP-HCN), thereby reducing "synaptic noise" and ensuring the selective strengthening of task-relevant neural signals. As a result, working memory processes, sustained attention, and impulse control are executed more effectively. Unlike stimulant medications, guanfacine modulates dopaminergic activity indirectly, thus exhibiting a more stabilizing pharmacological profile with a lower risk of anxiety, irritability, or insomnia. Furthermore, it provides physiological composure, offering a significant advantage in cases presenting with difficulties in anger management and agitation.
Clinical Use and ObservationsRandomized controlled trials demonstrate that the drug provides significant improvements in behavioral regulation, attention span, and impulse control when used both as monotherapy and as an adjunct to stimulant treatments. In clinical practice, the addition of guanfacine can provide distinct behavioral tranquility and emotional stability in cases that show a partial response to stimulants or where treatment is limited by side effects. Sedation is generally mild and may be considered a compliance-enhancing feature in most cases. Cardiovascular side effects are rare when dose titration is performed carefully. In these respects, guanfacine stands out as a frequently preferred agent in clinical practice for child and adolescent cases targeting behavioral stability, serving as either a complementary or alternative option to stimulant therapy.
PHOSPHATIDYLSERINE AND CITICOLINE:
THE CONTRIBUTION OF NEUROCOGNITIVE PERFORMANCE
Dr. İpek Aksakal

In recent years, there has been growing interest in agents that support cognitive function in disorders such as ADHD, cognitive impairment, and Alzheimer's disease. In this context, Phosphatidylserine (PS) and Citicoline (CDP-choline) have attracted attention as non- pharmacological, cognition- supporting compounds with the potential to enhance cognitive performance.
What is Phosphatidylserine?
Phosphatidylserine is a phospholipid which plays a pivotal role in synaptic plasticity, cell membrane integrity, and neuronal communication. It is defined as a negatively charged lipid membrane, approximately 2–15% of all membrane phospholipids. It is found in various organs, particularly in the central nervous system. The highest concentration of phosphatidylserine is found in the brain, approximately 13–15% of total brain phospholipids. PS are also found in heart muscle tissue, where it contributes to energy metabolism. This distribution reveals that phosphatidylserine is primarily involved in neurological functions and cellular structures with high energy requirements.
Its dense presence in the membrane structure of nerve cells indicates that Phosphatidylserine plays a critical role in cognitive functions, synaptic transmission, and cellular energy production.
Phosphatidylserine plays a significant role in synaptic transmission by regulating the primarily release of noradrenaline, acetylcholine, serotonin, and dopamine and the receptor functions associated with these systems.
It contributes to the preservation of neuronal network integrity by supporting intercellular communication and interaction.
Furthermore, it undertakes critical functions in the development and plasticity of the central nervous system by participating in neuronal survival, neurite outgrowth, and synaptogenesis processes.
In cells undergoing apoptosis, PS is translocated to the outer surface of the cell membrane, functioning as an "eat me" signal recognized by macrophages, ensuring that phagocytosis occurs effectively. In addition to contributing to the neuronal message transmission capacity in brain cells, it provides a protective effect against oxidative stress by exhibiting antioxidant properties at the cellular level.
Dosage and Administration
It can be administered in divided doses, morning and evening. The ideal dose is 150–200 mg/day, and it is recommended to be taken with food to facilitate absorption.
The FDA has validated that a daily dose of 300 mg is safe for use for up to 6 months. It has been stated that Phosphatidylserine is proven safe in sensitive groups and healthy children. In adults, the daily dose may be increased up to 400 mg.
Orally administered (PS) has high bioavailability in humans. Following absorption, it can easily cross the blood-brain barrier to access the central nervous system.
A meta-analysis published in 2022 evaluated the effect of phosphatidylserine (PS) supplementation on cognitive functions in elderly individuals.
Participants were administered 100– 300 mg of PS daily for periods ranging from 6 weeks to 6 months, and significant improvements were observed, particularly in the domain of short-term memory. It was reported that PS slowed cognitive decline and enhanced cognitive performance in individuals with mild cognitive impairment. In a study conducted on Major Depressive Disorder, the group receiving citicoline showed higher remission rates and a more rapid and significant reduction in Hamilton Depression Rating Scale scores compared to the placebo group. In a group of healthy adolescents aged 13–19 years, citicoline supplementation administered at doses of 250 mg and 500 mg for 28 days resulted in significant improvements in attention tests and motor speed. In another study involving healthy adults, participants were given a caffeine-based beverage containing 250 mg of citicoline. Continuous Performance Test results showed faster reaction times and better visuospatial processing in the citicoline-caffeine group. EEG analyses observed significant increases in P450 waves related to working memory and attention. These findings suggest that citicoline may positively affect cognitive functions. Both compounds possess pharmacological neuroprotective potential and stand out with a safe usage profile in children and adolescents.
INTEGRITY OF BLINDING IN PSYCHIATRIC RANDOMIZED CONTROLLED TRIALS:
Dr. Ece Pabuşçu

Randomized controlled trials (RCTs) particularly when designed as double-blind studies are considered the gold standard for evaluating the efficacy of psychiatric treatments because they minimize systematic biases. The purpose of blinding is to ensure that participants, outcome assessors, and treatment providers do not know which group a participant is assigned to, thereby reducing expectancy effects, observer bias, and performance bias.
In double-blind RCTs, preserving internal validity depends on maintaining blinding from the start of the trial through outcome analysis. Adverse drug effects, participants’ prior expectations, or perceived treatment efficacy can compromise the integrity of blinding.
Consequently, even when studies are labeled “double-blind,” the success of blinding may remain uncertain. Loss of blinding increases the risk of bias, particularly in psychiatry where self- report measures are frequently used. Blinding integrity is typically tested by asking participants and/or assessors to guess which treatment arm they believe they were in.
In trials where blinding is compromised, treatment effects may be reported as larger than they truly are. This indicates, when blinding cannot be maintained, expectancy effects and observer bias may distort outcomes.
Responses are usually collected as “active treatment,” “placebo,” or “don’t know,” and analyzed using methods such as the Bang Blinding Index (BI) or Cohen’s kappa coefficient.
Recent systematic reviews suggest that blinding integrity is rarely assessed in psychiatric RCTs. An important distinction in unblinding is between benign unblinding (blinding is broken because the treatment effect is genuinely strong) and malicious unblinding (blinding is broken due to side effects or treatment-specific cues).
Whereas benign unblinding may not threaten trial validity, malicious unblinding can introduce substantial bias in estimates of efficacy and tolerability. To make this distinction, it is necessary to evaluate whether blinding failed and, crucially, why it failed.
Critiques of Assessing Blinding Integrity
Determining blinding integrity in psychiatric RCTs is methodologically and ethically debated. The concept of equipoise refers to a genuine uncertainty on the part of the researcher that either treatment arm could be equally beneficial. However, asking participants or assessors to guess treatment allocation may undermine this balance; side effects or perceived efficacy can amplify expectancy responses and observer bias. When the rationale behind guesses is not elicited, it becomes difficult to understand why blinding was compromised. Moreover, testing blinding only at the end of the study may miss changes occurring during the trial.
In summary, the very methods used to assess blinding can themselves contribute to the erosion of blinding.
In psychiatric RCTs, assessment of blinding integrity is frequently neglected. Reported rates of blinding assessment range from approximately 1% to 7%. When blinding is tested, unblinding is often observed in active-treatment arms, whereas blinding tends to be better preserved in placebo arms.
Assessing blinding integrity is not merely a methodological detail; it is critical for the validity and reliability of trial findings. At the same time, the process of assessing blinding poses its own challenges: equipoise may be disrupted, and the clinical meaning of certain blinding metrics is not always clear.
Accordingly, the following recommendations are emphasized for future research:
- Blinding assessment should be conducted at multiple stages throughout the trial, not only at the end.
- Participants’ and assessors’ guesses should be accompanied by inquiry into how they arrived at their judgments, and benign versus malicious unblinding should be distinguished.
- Assessments should be conducted by independent personnel or via anonymous questionnaires to reduce expectancy effects.
- Standardized, comparable methods should be used, and their clinical implications should be explicitly discussed.
A Profound Loss for the Neuroscience Community:
Prof. Dr. Dan J. Stein
With great sorrow we mark the loss of Dan J. Stein (1962–6 December 2025), a towering figure in contemporary psychiatry and mental-health research. Over the course of a distinguished career, Professor Stein held the position of Chair of the Department of Psychiatry and Mental Health at University of Cape Town (UCT), and served as Director of the South African Medical Research Council (MRC) Unit on Risk & Resilience in Mental Disorders.
Educated in medicine at UCT, followed by psychiatry training and a post-doctoral fellowship in psychopharmacology at Columbia University, Stein went on to earn a PhD in clinical neuroscience and a DPhil in philosophy from Stellenbosch University — a rare and powerful combination of science and humanities informing his work.
Across basic neuroscience, clinical research, and public-health epidemiology, Stein’s scholarship focused particularly on anxiety disorders, obsessive-compulsive and related disorders, and trauma- and stress-related conditions.
He played a foundational role in shaping modern diagnostic frameworks, contributing to both the revision processes for DSM 5 and ICD 11 work-groups on obsessive-compulsive and related disorders.
His publication record is prodigious: hundreds of peer-reviewed papers, dozens of edited volumes (among them the influential book Problems of Living: Perspectives from Philosophy, Psychiatry, and Cognitive Affective Science), and mentoring that has shaped a new generation of global mental-health scientists.
Stein’s work earned him top honors: including the Max Hamilton Memorial Award for contributions to psychopharmacology, the MRC “Platinum Award,” and the World Federation of Societies of Biological Psychiatry Lifetime Achievement Award. Because he bridged neurobiology, clinical care and philosophy; his passing is a profound loss. But his legacy lives on: in his writings, in the many students and colleagues he inspired, and in the lives of countless patients helped by his work.

